Table of VSEPR Shapes
Valence Shell Electron Pair Repulsion model shapes, listed
by number of bonds. Rows are colored according to the total
number of electron domains.
|
Ā
|
Number of Bonds on Central Atom
|
|
1
|
2
|
3
|
4
|
5
|
6
|
Number of
Lone Pairs on
Central Atom
|
0
|
Linear
|
Linear
|
Trigonal
Planar
|
Tetrahedral
|
Trigonal
Bipyramidal
|
Octahedral
|
|
1
|
Linear
|
Bent
|
Trigonal Pyramidal
|
See-saw
|
Square Pyramidal
|
|
2
|
Linear
|
Bent
|
T-Shaped
|
Square Planar
|
|
3
|
Linear
|
Linear
|
Bonding
Electron
Domains
|
Lone
Pair
Domains
|
Total
Electron
Domains
|
Shape
|
Ideal
Bond
Angle(s)
|
Examples
|
Image
|
|
1
|
0, 1, 2, or 3
|
1, 2, 3, or 4
|
linear
|
none
|
H2,
N2,
O2,
CO, CN–
|
 
|
|
2
|
0
|
2
|
linear
|
180°
|
BeCl2,
CO2
|
 
|
|
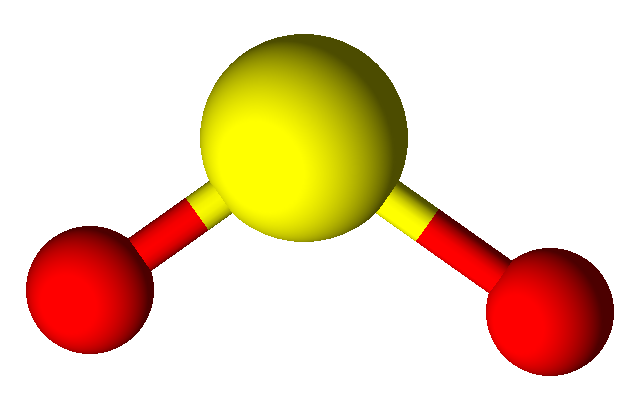
2
|
1
|
3
|
bent
|
120°
|
SO2,
H2CO,
NO2–
|
|
|
2
|
2
|
4
|
bent
|
109.5°
|
H2O,
SCl2,
NH2–
|
 
|
|
2
|
3
|
5
|
linear
|
180°
|
XeF2
|
 
|
|
3
|
0
|
3
|
trigonal planar
|
120°
|
BF3,
AlCl3,
CO32–,
NO3–
|
 
|
|
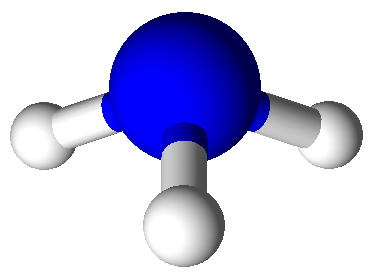
3
|
1
|
4
|
trigonal pyramidal
|
109.5°
|
NH3,
PCl3,
ClO3–,
H3O+
|
|
|
3
|
2
|
5
|
T-shaped
|
90°, 180°
|
ClF3,
BrF3
|
 
|
|
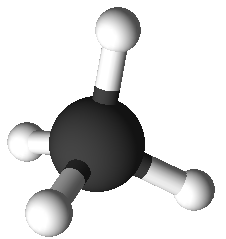
4
|
0
|
4
|
tetrahedral
|
109.5°
|
CH4,
SiCl4,
ClO4–,
SO42–
|

|
|
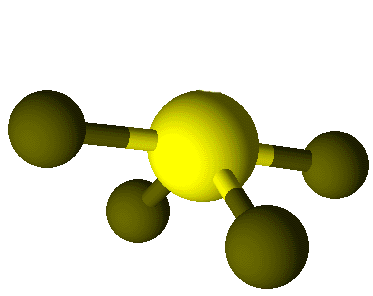
4
|
1
|
5
|
see-saw
|
180°, 120°
|
SF4,
SeF4,
IO2F2–,
ClF4+
|

|
|
4
|
2
|
6
|
square planar
|
90°
|
XeF4,
PtCl42–
|


|
|
5
|
0
|
5
|
trigonal bipyramidal
|
90°, 120°
|
PCl5,
PF3Cl2,
SbF5
|
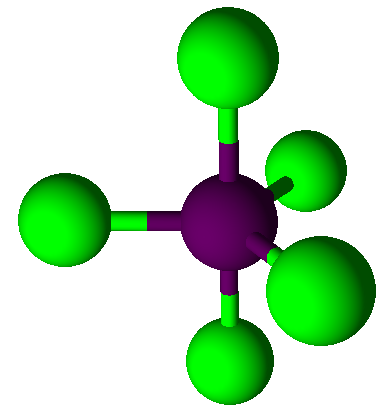

|
|
5
|
1
|
6
|
square pyramidal
|
90°
|
IF5,
BrF5,
XeOF4
|


|
|
6
|
0
|
6
|
octahedral
|
90°
|
SF6,
Fe(CN)63–
|


|
page break
Bonding
Electron
Domains
|
Lone
Pair
Domains
|
Total
Electron
Domains
|
Shape
|
Ideal
Bond
Angle(s)
|
Examples
|
Image
|
|
1
|
0, 1, 2, or 3
|
1, 2, 3, or 4
|
linear
|
none
|
H2,
N2,
O2,
CO, CN–
|
 
|
|
2
|
0
|
2
|
linear
|
180°
|
BeCl2,
CO2
|
 
|
|
3
|
0
|
3
|
trigonal planar
|
120°
|
BF3,
AlCl3,
CO32–,
NO3–
|
 
|
|
2
|
1
|
3
|
bent
|
120°
|
SO2,
H2CO,
NO2–
|
 
|
|
4
|
0
|
4
|
tetrahedral
|
109.5°
|
CH4,
SiCl4,
ClO4–,
SO42–
|


|
|
3
|
1
|
4
|
trigonal pyramidal
|
109.5°
|
NH3,
PCl3,
ClO3–,
H3O+
|
 
|
|
2
|
2
|
4
|
bent
|
109.5°
|
H2O,
SCl2,
NH2–
|
 
|
|
5
|
0
|
5
|
trigonal bipyramidal
|
90°, 120°
|
PCl5,
PF3Cl2,
SbF5
|


|
|
4
|
1
|
5
|
see-saw
|
180°, 120°
|
SF4,
SeF4,
IO2F2–,
ClF4+
|


|
|
3
|
2
|
5
|
T-shaped
|
90°, 180°
|
ClF3,
BrF3
|
 
|
|
2
|
3
|
5
|
linear
|
180°
|
XeF2
|
 
|
|
6
|
0
|
6
|
octahedral
|
90°
|
SF6,
Fe(CN)63–
|


|
|
5
|
1
|
6
|
square pyramidal
|
90°
|
IF5,
BrF5,
XeOF4
|


|
|
4
|
2
|
6
|
square planar
|
90°
|
XeF4,
PtCl42–
|


|











